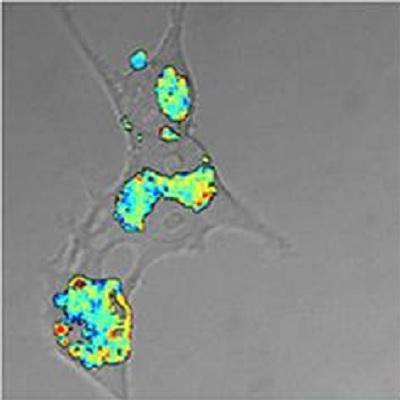
New research by the Laser Analytics Group sheds light on the protein aggregation reactions at the heart of Alzheimer's disease.
Research led by Dr. Gabi Kaminski Schierle, and recently published in Chemistry and Biology, reveals how Amyloid-β, a protein involved in Alzheimer's disease, develops into a pathogenic species. The group has developed a fluorescent sensor concept, which makes it possible to study how proteins misfold and aggregate in living cells. Crucially it was shown that the kinetics of Aβ aggregation are vastly different in brain cells, than in the test tube, and further that the most pathogenic forms of Amyloid-β aggregate much faster in live cells than had previously been assumed from corresponding studies in test tubes. The technique makes it possible to correlate the appearance of certain aggregate species with their gain of toxic function thus providing a tool to screen for potential therapeutic agents in more efficient ways than hitherto possible.
Read more on this research in a press release on Alzforum.
